Under the support of National Natural Science Foundation of China (Project No. 21373025, 91544227), research team of Professor Xiuhui Zhang at Beijing Institute of Technology, in cooperation with group at University of Nebraska-Lincoln led by Professors Xiao Cheng Zeng and Joseph S. Francisco, group at Institute of Chemistry of the Chinese Academy of Sciences led by Professor Maofa Ge, and group at the University of Helsinki, made important breakthrough in the study of new particle formation in highly polluted areas. Research results were published with the title of “Self-Catalytic Reaction of SO3 and NH3 to Produce Sulfamic Acid and Its Implication to Atmospheric Particle Formation” in Journal of the American Chemical Society on August 8, 2018. The website link is: http://pubs.acs.org/doi/abs/10.1021/jacs.8b04928.
Aerosol new particles are an important source of particulate matter and cloud condensation nuclei in the atmosphere, which have significant impacts on environmental quality, regional climate and human health. New particle formation (NPF) events can occur more frequently in Chinese megacities because of higher-level air pollution. Nucleation is the first important stage of the NPF. However, due to limitation of the experimental techniques, the molecular-level mechanism of nucleation, especially that related to the realistic pollution events, is still not fully understood. Therefore, it is of special importance in atmospheric science to achieve improved understanding of the mechanism underlying nucleation of new particles, and to assess the effects of new particles on environment and health.
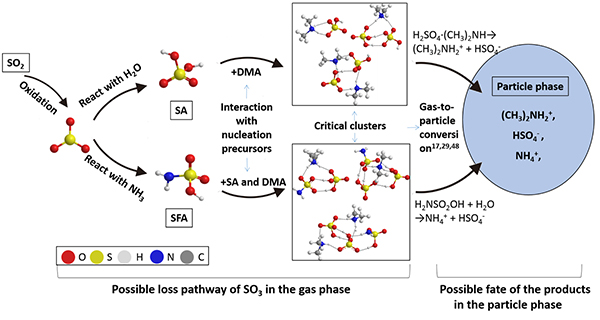
In this study, the self-catalytic reaction between SO3 and NH3 is investigated by using both quantum chemical calculation and the atmospheric cluster dynamics model. The research team identifies a new and competitive loss pathway of SO3 when abundant ammonia (NH3) species are available in the atmosphere. Specifically, the reaction between SO3 with NH3, which produces sulfamic acid, can be self-catalyzed by the reactant (NH3). Hence, in dry and highly polluted regions with relatively high concentration of NH3, the effective rate constant for the bimolecular SO3-NH3 reaction can be sufficiently high such that the new loss pathway for SO3 becomes as competitive as the conventional loss pathway of SO3 with water. The final product of the self-catalyzed reaction, sulfamic acid, can subsequently participate in NPF, thereby enhancing the fastest possible rate of NPF resulted from sulfuric acid and dimethylamine by about a factor of two. The new chemical mechanism has implication to improve atmospheric modeling of NPF especially for events occurring in highly polluted regions in China.

Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@donnasnhdiary.org
京ICP备05002826号 文保网安备1101080035号 Copyright 2017 NSFC, All Right Reserved